For Doctors

Osteoporosis is a skeletal disorder characterized by bone loss, low bone mass, and structural degradation of bone tissue, especially trabecular bone, yielding attenuated bone strength that, in turn, increases the risk of fracture. Bone strength reflects not only changes in bone density, but also in bone quality-which encompasses bone architecture, the presence or absence of microfractures, mineralization, and bone turnover.
The population of India is expected to increase to 1,613 million by 2050, of which 19.6% (315 million), will be adults over 60 years. This indicates that in the years to come, a very large population will be at risk for osteoporosis, especially with the increase in the elderly population. Indians have a lower BMD compared to Western countries and studies suggest that 20% of women and about 10-15% of men are osteoporotic in India. Osteoporotic fractures are common in both sexes, and may occur at a younger age than in the western countries.
The National Osteoporosis Foundation (NOF) estimates that, in the United States, 10 million individuals, 80% of whom are women, already have osteoporosis, with an additional 34 million individuals at risk because of low bone mass.1,2 The prevalence of low bone density increases dramatically with age, affecting 37% of women between the ages of 50 and 59 years, 50% between the ages of 60 and 69 years, 75% between 70 and 79 years, and 87% of women over age 80.2 The lifetime fracture risk of a patient with osteoporosis is as high as 40%, and fractures most commonly occur in the spine, hip, or wrist, but other bones such as the trochanter, humerus, or ribs can also be affected. From a patient’s perspective, a fracture and the subsequent loss of mobility and autonomy often represent a major drop in quality of life. Although the majority of research on osteoporosis in the past has focused on women, osteoporosis is also becoming an increasingly important problem in men. One in 8 men ≥50 yrs of age will have an osteoporosis-related fracture, and this figure is predicted to rise with an aging male population. Although fragility fractures in men occur an average of 10 years later compared with those in women5, most clinical fractures result in greater morbidity and mortality.
Reversible Lifestyle Factors
- Low calcium intake
- Immobilization
- Low body mass index
- High salt, cola, or caffeine intake
- Vitamin D insufficiency
- Inadequate physical activity
- Smoking
- Alcohol (>3 drinks/day)
Rheumatic and Autoimmune Diseases
- Rheumatoid arthritis
- Lupus
- Ankylosing sponylitis
Hypogonadal States
- Androgen insensitivity
- Hyperprolactinemia
- Ovarian failure
Liver Diseases
- Autoimmune hepatitis
- Alcoholic cirrhosis
Endocrine Disorders
- Adrenal insufficiency
- Hyperparathyroidism
- Type 1 diabetes
Cushing’s syndrome - Hypothyroidism
Gastrointestinal Disorders
- Celiac disease
- Inflammatory bowel disease
- GI surgery, gastric bypass
Medications
- Anticoagulants (heparin)
- Anticonvulsants
Immunosuppressants - Lithium
- Glucocorticoids (>5mg/d prednisone or equivalent for >3mo)
- Aromatase inhibitors
- Barbiturates
- Gonadotropin-releasing hormone agonists
Measurement of bone mineral density (BMD) by dual-energy x-ray absorptiometry (DXA) is the most widely used method of diagnosing osteoporosis. Hip, spine, or wrist BMD is expressed as a T-score, the number of standard deviations (SD) above (+) or below (-) the young adult mean value. The World Health Organization” classifies bone mineral density on the basis of this T-score (Table 2).

DXA technology can measure BMD at virtually any skeletal site, but clinical use has been concentrated on the lumbar spine, proximal femur, forearm and total body. Although other factors, such as trabecular bone structure, are important, central BMD measurements via DXA are helpful in the diagnosis of osteoporosis for estimating the risk of nontraumatic fracture and in choosing and monitoring treatments.”
The National Osteoporosis Foundation (NOF) recommends testing of all women age 65 and older. NOF also provides indications for the use of DXA in other patient populations as well.

- Hip or vertebral (clinical or morphometric) fracture.
- T-score of -2.5 or less at the femoral neck or spine after appropriate evaluation to exclude secondary causes of osteoporosis
- Low bone mass (T-score between-1.0 and -2.5 at the femoral neck or spine) and a 10-year probability of a hip fracture of 3% or greater OR a 10-year probability of major osteoporosis-related fracture of 20% or greater based on the US-adapted World Health Organization FRAX” algorithm.
- The most recent American Association of Clinical Endocrinologists (AACE) postmenopausal osteoporosis guidelines endorse the NOF recommendations for initiation of treatment in postmenopausal women.”

- The native hormone secreted by the parathyroid gland chief cell is human parathyroid hormone (hPTH (1-84)], a single chain polypeptide with 84 amino acids which plays a central role in the maintenance of calcium and phosphate homeostasis in mammals.
- The ambient extracellular calcium level signals an increase in PTH secretion in response to a decrease in calcium concentration via the calcium-sensing receptors on the parathyroid cellular membrane.
- PTH acts directly to increase renal tubular calcium reabsorption and indirectly to enhance intestinal calcium absorption via its stimulatory action on renal 1-cholecalciferol hydroxylase (thereby increasing circulating calcitriol).
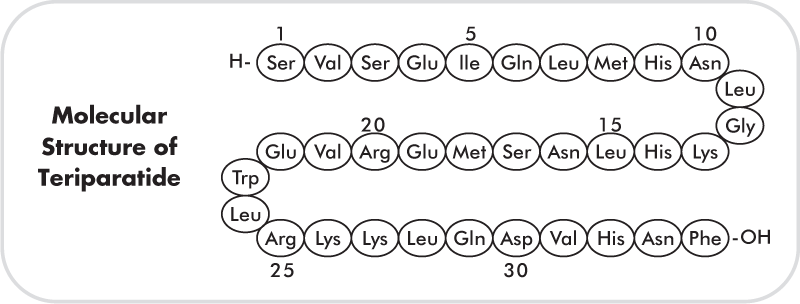
- The knowledge of the molecular structure of PTH allowed the production of hPTH (1-84) for treatment in humans. Moreover, the discovery that the N-terminal 34 amino acid portion of the native PTH molecule could fully activate the PTH/PTHrP (parathormone-related peptide analogue) receptor, has led to the generation of pharmacological products comprising only this portion, such as hPTH (1-34) and recombinant human PTH [hPTH (1-34)) (Figure 3). Teriparatide is the generic name for all PTH (1-34) molecules. It has a molecular weight of 4117.8 daltons and is manufactured using a strain of Escherichia coli modified by recombinant DNA technology.
- Teriparatide is an anabolic therapy that improves bone density, reduces vertebral and non-vertebral fracture incidence, improves the geometric and microarchitectural properties of bone, and has been observed to accelerate fracture healing.
- It is administered subcutaneously using a pen delivery device, has favourable pharmacokinetics, and is approved for the treatment of postmenopausal osteoporosis, osteoporosis in men, and glucocorticoid-induced osteoporosis.
- It should be considered as first-line therapy in patients at high risk for fracture, or in patients for whom the physician is not satisfied with the effectiveness of other registered therapies. Patients who experience a fracture on antiresorptive therapy, and treatment-naïve patients with prevalent fractures are eligible for teriparatide therapy.
- Overall, teriparatide is well-tolerated and safe in most patients. However, administration beyond 2 years is not recommended owing to insufficient clinical data beyond this time point.
- Teriparatide exerts its anabolic effect, at least in part, by modulating the differentiation, proliferation, and activity of osteoblast pool by
- (A) Increasing the development of the osteoblast
- (B) Inhibiting osteoblast apoptosis and increasing their life span
- (C) Reactivating lining cells to resume their matrix synthesizing function.
- Teriparatide exerts anti-mitotic effects on replicating osteoblast progenitors, and may also inhibit their apoptosis. The anti-mitotic effects may be necessary for differentiation in response to locally produced autocrine/paracrine growth factors (TGF, FGF, IGF) regulated by PTH, as well as factors released from the bone matrix during bone resorption. Teriparatide may also increase the number of osteoblast progenitors by preventing the differentiation of adipocytes from pleuripotent progenitors.
- Teriparatide stimulates bone formation through an increase in the bone remodeling rate with bone formation favored over bone resorption, thereby resulting in a net gain of bone deposited in each basic multicellular unit (BMU).
- Once-daily administration of teriparatide stimulates new bone formation on trabecular and cortical (periosteal and/or endosteal) bone surfaces by preferential stimulation of osteoblastic activity over osteoclastic activity. This cumulatively manifests as an increase in skeletal mass, an increase in markers of bone formation and resorption, and an increase in bone strength.
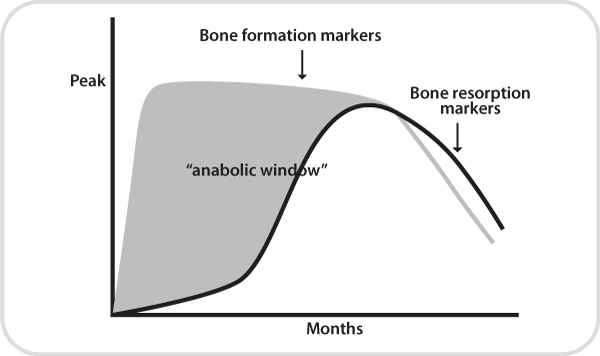
- Clinical studies have shown that teriparatide induces large increases in biochemical markers of bone formation after one month of therapy followed by a delayed increase in bone resorption markers. This difference in the rise of bone markers produces what has been termed “the anabolic window”, a period when bone formation is greater than bone resorption,” The anabolic window is based on the bone marker response to teriparatide, and is the time of its maximum anabolic effect. The anabolic window also has implications for monitoring of patients who are receiving osteoporosis pharmacotherapy with teriparatide.”
- Treatment of postmenopausal women with osteoporosis at high risk for fracture (a prior history of osteoporotic fracture, multiple risk factors for fracture), or patients who have failed or are intolerant to other available osteoporosis therapies.
- Increase of bone mass in men with primary or hypogonadal osteoporosis at high risk for fracture, or patients who have failed or are intolerant other available osteoporosis therapies.
- Treatment in men and women with osteoporosis associated sustained systemic glucocorticoid therapy (daily dosage equivalent and women >5 mg of prednisone) at high risk for fracture, or patients who have failed or are intolerant to other available osteoporosis therapies.
- Gemfrac (teriparatide) is supplied as a ,sterile, colorless, clear, isotonic solution in a glass cartridge with Gemfrac delivery prefilled pen devise only for subcutaneous use only.
- Each Gemfrac glass cartridge contains 600 ug of teriparatide in 2.4 ml.
- Each cartridge should be inserted into a delivery device to deliver 20 ug of teriparatide per dose each day for up to a 28 day period. The recommended dose for Gemfrac (teriparatide) is 20 µg subcutaneously once a day into the thigh or abdominal wall. Gemfrac (teriparatide) is administered via a multi-dose prefilled pen delivery device containing 28 daily doses of 20g. It should be administered initially under circumstances in which the patient can sit or lie down if symptoms of orthostatic hypotension occur.
- The delivery device should be stored under refrigeration at 2 to 8°C (36 to 46°F) at all times.
- While the delivery device is in use, the time out of the refrigerator should be minimized, the dose may be delivered immediately following removal from the refrigerator
- It is important to recap the delivery device when not in use to protect the cartridge from physical damage and light.
- The absolute bioavailability is approximately 95% based on pooled data from 20, 40, and 80 ug doses. Rates of absorption and elimination are rapid.
- Reaches peak serum concentrations about 30 minutes after subcutaneous injection of a 20 ug dose, and declines to non quantifiable concentrations within 3 hours. The half-life in serum is approximately 1 hour.
- Systemic clearance of teriparatide (approximately 62 L/hr in women and 94 L/hr in men) exceeds the rate of normal liver plasma flow consistent with both hepatic and extra-hepatic clearance. No metabolism or excretion studies have been performed with teriparatide. However, its peripheral metabolism is believed to occur by non-specific enzymatic mechanisms in the liver folloawed by excretion via the kidneys.
- Pharmacokinetic data in pediatric patients are not available No age-related differences in teriparatide pharmacokinetics have been detected (range 31 to 85 years).
- Although systemic exposure to teriparatide is approximately 20% to 30% lower in men than women, the recommended dose for both genders is 20 g/day.
- The influence of race on pharmacokinetic parameters has not been determined.
- No pharmacokinetic differences have been identified in patients with mild or moderate renal impairment [creatinine clearance (CrCl) of 30 to 72 mL/min] administered a single dose of teriparatide.
- Elevations in the AUC and T½ of teriparatide have been observed in patients with severe renal impairment (CrCl<30 mL/min). However, the maximum serum concentration of teriparatide has not been seen to increase.
- No studies have been performed in patients undergoing dialysis for chronic renal failure. No studies have been performed in patients with hepatic impairment. Non-specific proteolytic enzymesin the liver (possibly from Kupffer cells) cleave teriparatide into fragments that are cleared from the circulation mainly by the kidney.
- Sporadic case reports have suggested that hypercalcemia may predispose patients to digitalis toxicity. Because teriparatide may transiently increase serum calcium, it should be used with caution in patients taking digoxin. • The 24-hour urinary excretion of calcium has been observed to be reduced by a clinically insignificant amount (15%) following coadministration of hydrochlorothiazide with teriparatide.
- Coadministration of intravenous furosemide (20 to 100 mg) with teriparatide 40 g may result in small, clinically insignificant increases in the serum calcium (2%) and 24-hour urinary calcium (37%).
- Clinical trial data has indicated significant benefits for teriparatide 20μg in comparison to placebo with regards to the following outcomes
- Reduction in the number of new vertebral fractures
- Reduction in the number of new non-vertebral fractures
- Increased lumbar and femoral BMD
- Improvement in bone microarchitecture
- Improvement in fracture healing
- It is contraindicated in patients with hypersensitivity to teriparatide or to any of its excipients.
- Patients with Paget’s disease of bone, bone metastases, history of skeletal malignancies, open epiphyses (pediatric and young adult patients), and prior external beam/implant radiation involving the skeleton should not be treated with teriparatide
- It should not be prescribed for patients at increased baseline risk for osteosarcoma (e.g., Paget’s disease of bone or unexplained elevations of alkaline phosphatise; pediatric and young adult patients with open epiphyses; patient with prior external beam or implant radiation therapy involving the skeleton).
- Patients with metabolic bone diseases other than osteoporosis or patients with hypercalcemic disorders should not be treated with teriparatide.
- Teriparatide may increase serum calcium, urinary calcium, and serum uric acid, and may exacerbate active or recent urolithiasis.
- Clinicians and patients need to be vigilant regarding the potential development of transient orthostatic hypotension while administering the initial doses of teriparatide.
- Teriparatide should not be used for more than 2 yrs owing to insufficient clinical data beyond this time point.
- In Rats, teriparatide caused an increase in the incidence of osteosarcoma, a malignant bone tumor Because of the uncertain relevance of the rat osteosarcoma finding to humans.
A) Pregnancy and lactation:
- There are no adequate and well-controlled studies of teriparatide in pregnant women.
- In animal studies, teriparatide has produced skeletal may on mations mild growth retardation and motor deficits in rodents, when administered in dose much higher than the equivalent human dose. Teriparatide should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- It is not known whether teriparatide is excreted in human milk: Because of the tumorigenic potential for teriparatide based on animal studies, a decision should be made whether to discontinue nursing or to discontinue the drug, considering the risk-benefit ratio for the mother.
B) Pediatric population:
- Safety and Efficacy of teriparatide has not been evaluated in the pediatric setting
- Teriparatide should not be prescribed in patients at an increased baseline risk of osteosarcoma i.e., pediatric and young adult patients with open epiphyses No overall differences have been observed in therapeutic response, efficacy, and safety for teriparatide for the geriatric vs the younger population. However, the potentially greater sensitivity of some older individuals cannot be ruled out.
- Incidents of overdose have not been reported in clinical trials-effects of overdose that might be expected include delayed hypercalcemia, orthostatic hypotension, nausea, vomiting, dizziness, and headache.


